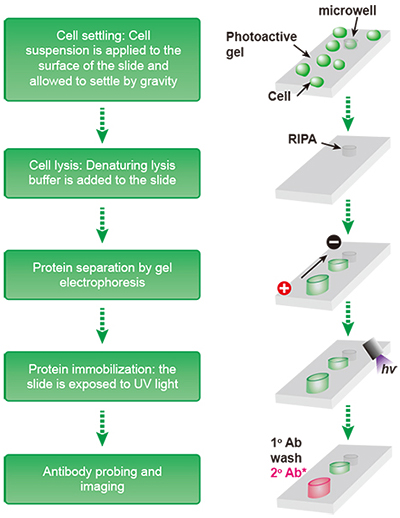
Cell lysis disrupts cell membranes and organelles, resulting in unregulated enzymatic activity that can reduce protein yield and lead to degraded proteins.To minimize sample variability, keep sample preparation workflows simple, and use reagents optimized for the specific sample type and target proteins.Image the blot using an appropriate imaging system with fluorescence detection mode.Place each blot in a sheet protector or on a clean surface prior to imaging to prevent contamination. Blots can be imaged immediately while still wet, or alternatively may be dried prior to imaging.Ensure the volume of the antibody solution is enough to fully cover the membrane and protect the membrane from bright light to prevent photobleaching of the fluorescent dyes. Incubate the membrane protein-side up in the secondary antibody solution for 1 hour with agitation at room temperature.1:20,000: 0.75 µL of secondary antibody in 15 mL wash buffer.1:10,000: 1.5 µL of secondary antibody in 15 mL wash buffer.1:5,000: 3 µL of secondary antibody in 15 mL wash buffer.Prepare dilutions of the conjugated secondary antibody to 0.4 to 0.1 µg/mL in appropriate volume of wash buffer or alternatively in blocking buffer.If using a fluorescently conjugated primary antibody, proceed to Step 11. Wash the membrane 3 times with agitation for 10 minutes each in wash buffer.Ensure the volume of the antibody solution is enough to fully cover the membrane. Incubate the membrane protein-side up in the primary antibody solution with agitation, for 1 hour at room temperature or overnight at 2–8☌.Dilute the primary antibody per supplier recommendations in the blocking buffer.Incubate the membrane with a sufficient volume of blocking buffer for 30–60 minutes at room temperature with agitation.After protein transfer, wash the membrane in deionized water 4 times for 5 minutes each with agitation to remove all transfer buffer.Follow manufacture instructions for wet, semi-dry, or dry transfer.Nitrocellulose: equilibrate directly in transfer buffer for 5 minutes.PVDF: pre-wet in methanol or ethanol (100%) for 30 seconds, briefly rinse in deionized water, and equilibrate in transfer buffer for 5 minutes.Follow manufacture instructions for dry membrane preparations. Prepare transfer membrane (semi-dry or wet transfers).Prepare transfer buffer for wet and semi-dry transfers based on gel chemistry.Image the blot using film or appropriate imaging system.Place the blot in clear plastic wrap or sheet protector and remove bubbles by rolling with blot roller or glass pipette.Remove the blot from working solution and drain excess reagent.when using high-performance substrates, such as SuperSignal substrates. when using standard ECL substrates or 5 min. Incubate the blot with the working solution for 1 min.Suggested volume of ~8–10 mL for mini blots and 15 mL for midi blots (0.1 mL working solution per cm 2 of membrane). Prepare working solution of chemiluminescent substrate based upon manufacture instruction.It is crucial to thoroughly wash the membrane at this step. Wash the membrane 6 times with agitation for 5 minutes each in wash buffer to remove any unbound secondary antibodies.

